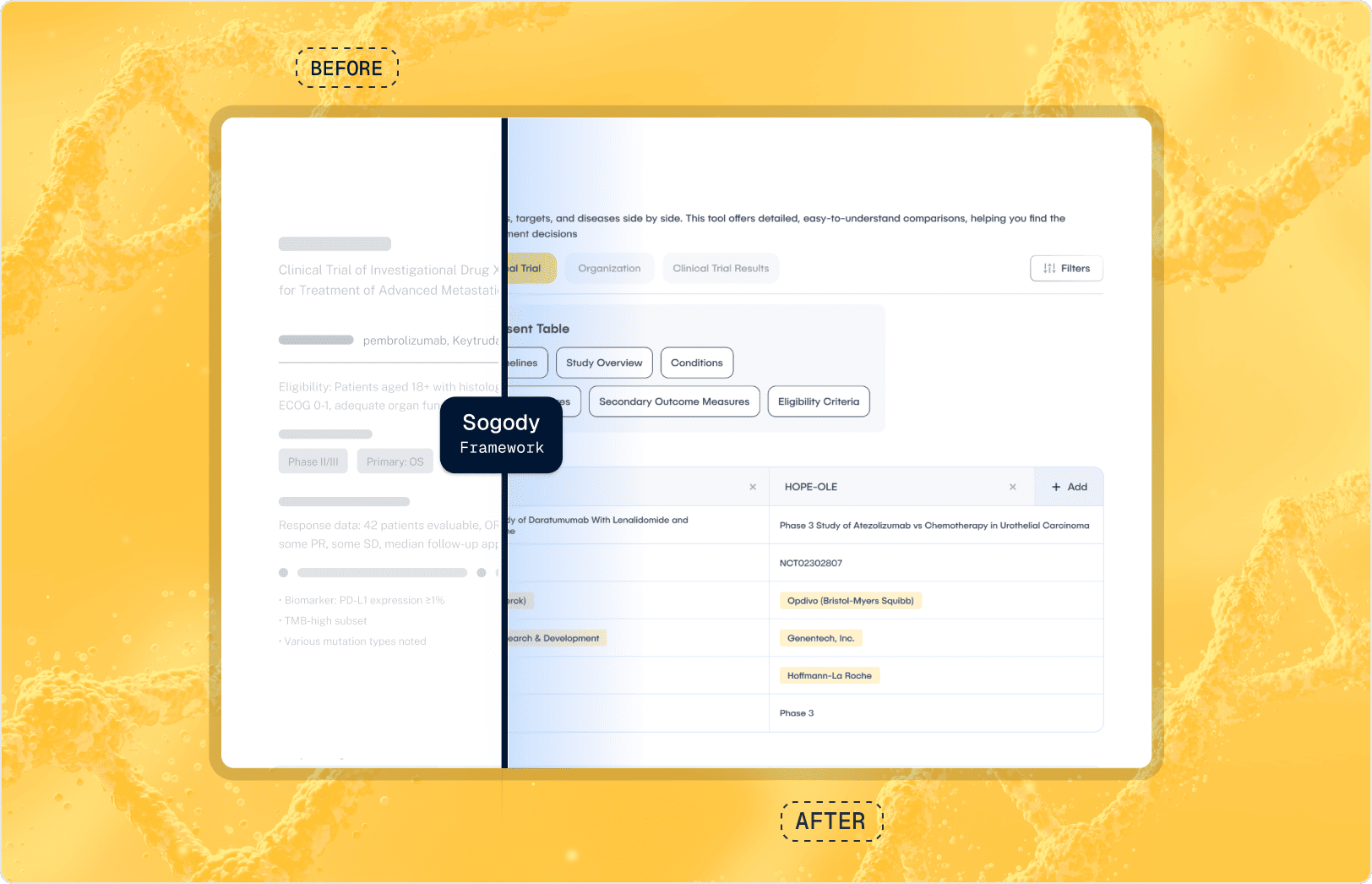
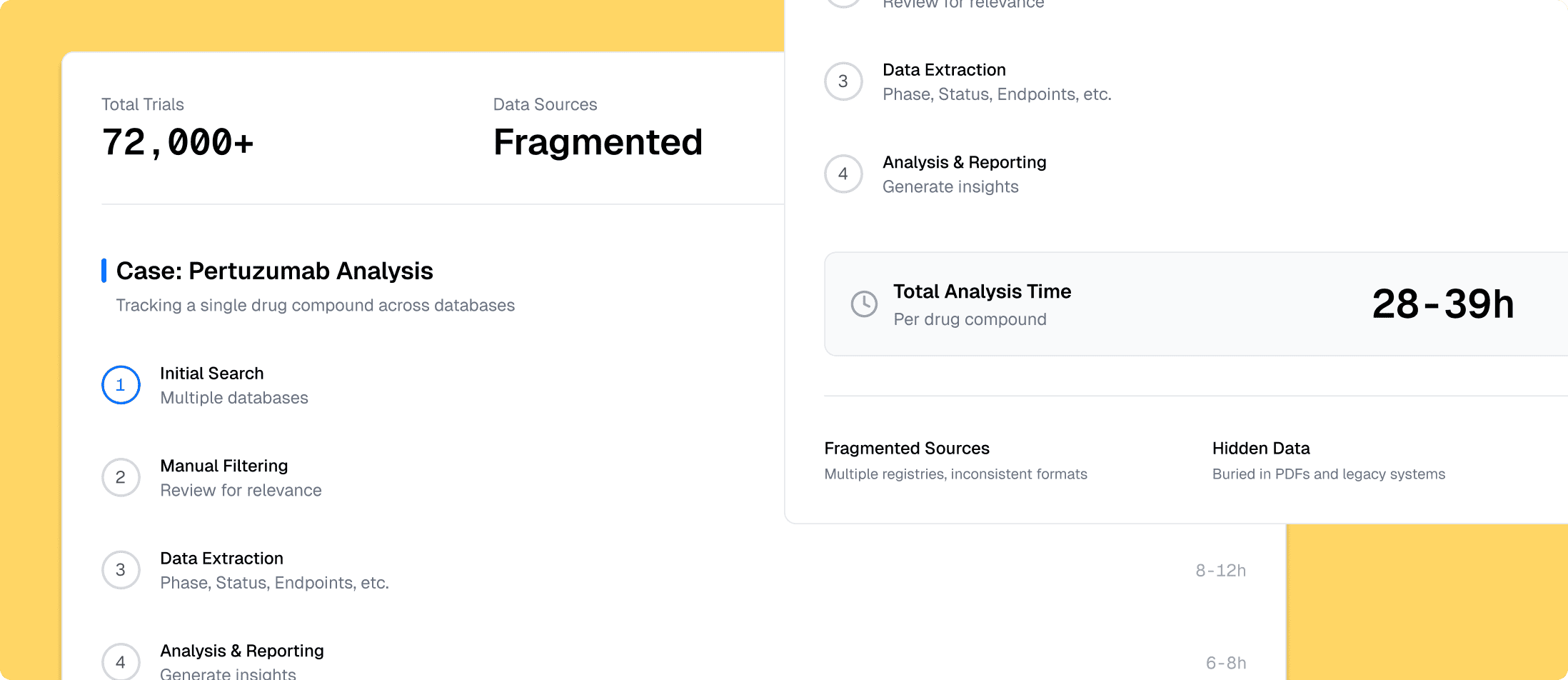
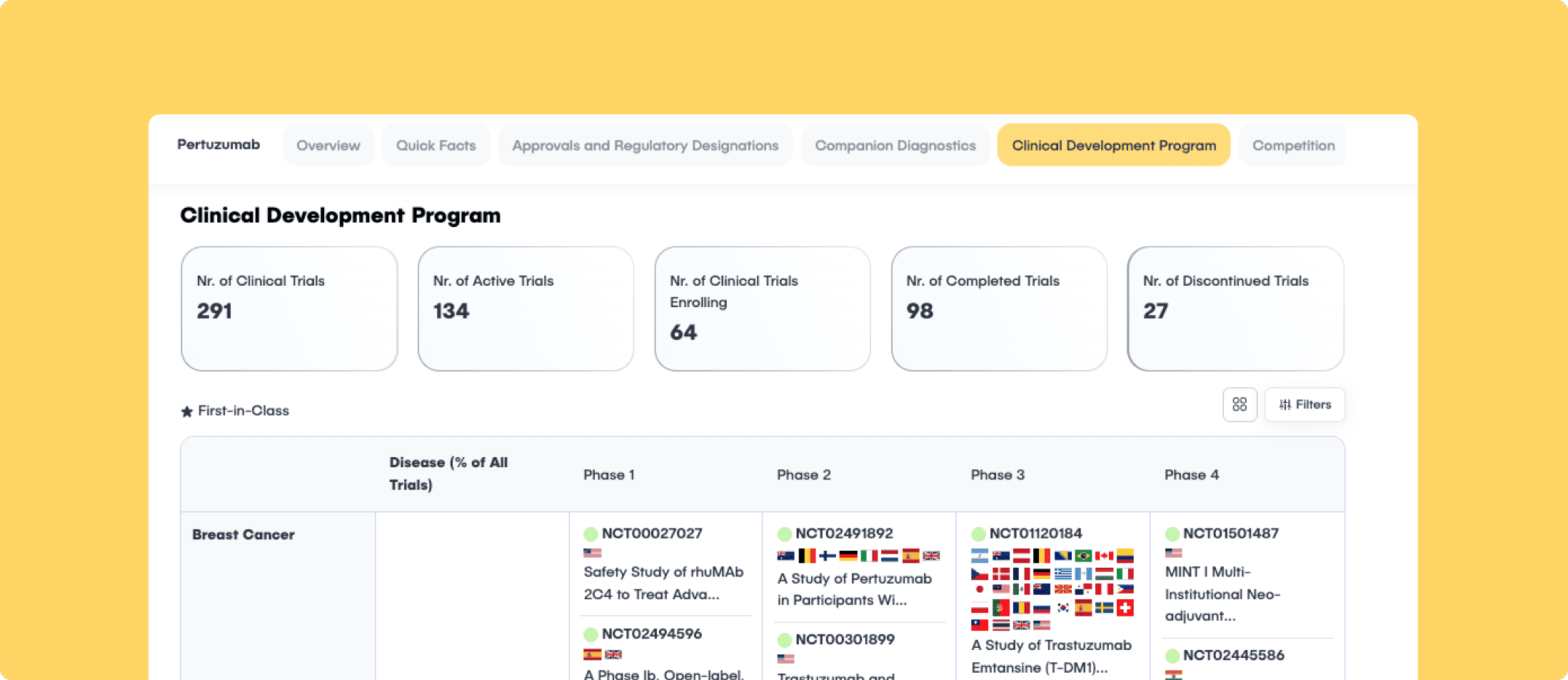
Source-Aware Ingestion (The Loading Dock)
- Instead of blindly reprocessing the entire database, intelligent agents diff version histories to detect semantically significant changes in trial text, ensuring only meaningful updates (not just typos) trigger the processing pipeline.
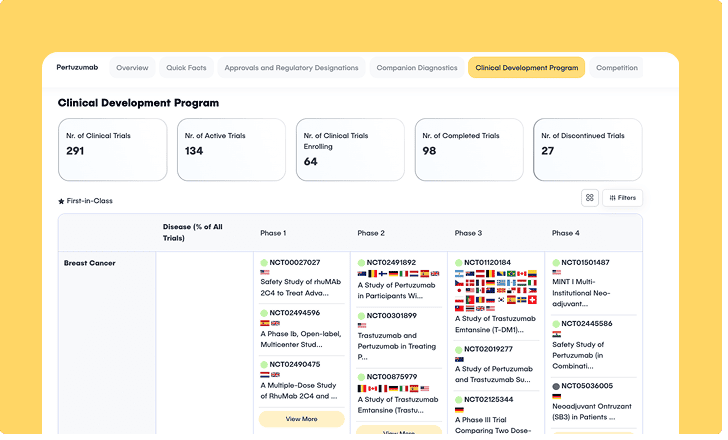
- Custom scrapers dynamically navigate anti-bot protections to routinely extract the thousands of specialized oncology trials from ChiCTR that other platforms miss.